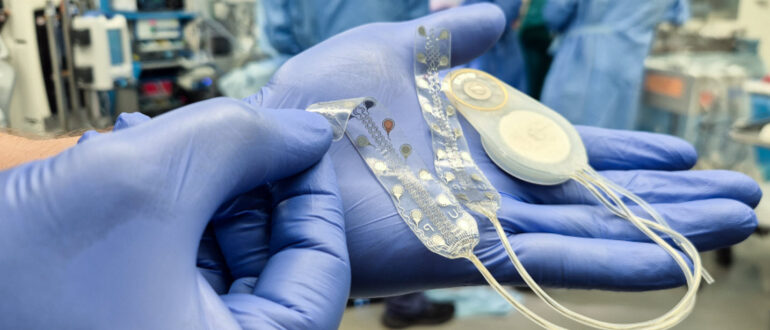
The CorTec Brain Interchange system’s neuroprosthetic is designed to function as a fully implanted wireless closed-loop platform that monitors and stimulates the brain simultaneously. You’ve probably seen various brain–computer interfaces in the news… but this specific hardware is unique because it avoids the need for external cables or bulky headgear that typically limits a survivor’s mobility during rehabilitation.
The techutilises ‘Air–Ray’ electrodes embedded within soft silicon sheets which are designed to rest upon the cortical surface rather than penetrating the brain tissue; this is a critical safety feature for long term use as it reduces the risk of inflammation or scarring whilst maintaining a high resolution for neural signal acquisition. In a notable clinical trial at the University of Washington, a participant was able to play the video game ‘Pong’ using nothing but his thoughts just two hours after the system was introduced. This speed of acquisition suggests that the decoding algorithms used to interpret your intent to move are becoming incredibly efficient. Dr Frank Desiere and his team have ensured the hardware remains identical for both the rehabilitative stimulation and the digital control aspects; so you’ve no requirement for additional surgical interventions or separate devices to switch between physical therapy and computer interaction.
The system has achieved an ‘FDA Breakthrough Device Designation’ in the United States… which accelerates the regulatory path for technologies that provide more effective treatment of life–threatening or irreversibly debilitating conditions. Prof Jeffrey Herron and other leading researchers involved in the study believe the ability to record and stimulate in real time allows for a more tailored approach to neuroplasticity. By delivering precisely timed electrical pulses… the device aims to strengthen the neural pathways you’ve lost after a stroke; essentially helping the brain to rewire itself more effectively than through traditional passive exercises alone. Although the primary trials are taking place across the Atlantic… the German engineering firm CorTec is actively looking toward the European and British markets for future routine implementation.
It’s difficult to pin down a precise date for when you might see this in a standard UK clinical setting; however the progression of clinical programmes for paralysis and the move toward more personalised neurotherapies suggest we are looking at a timeframe of several years rather than decades. The transition from feasibility studies to widespread NHS availability will depend heavily on larger scale trials proving long term efficacy and cost-effectiveness. ARNI Stroke Rehab Ul says that the dual capability of brain stimulation and digital control mean that closed–loop neurotherapies could possibly be moving closer to becoming a standard part of long term recovery in the future, but whilst the technology is promising, there’s currently no firm date for when such advanced systems will be available for routine use in the UK. Interesting however to know that this tech is out there and hopefully coming our way….